Lighting the future with efficient renewable energy
with Prof. Akari Hayashi
Ever since the pre-industrial revolution, global temperatures have been steadily rising, primarily due to the combustion of fossil fuels that release heat-trapping greenhouse gases—a majority in the form of carbon dioxide—into our atmosphere. Without action, this continued global warming will lead to increased frequency of extreme weather events and untold damage to our already strained ecosystem in what has been now dubbed a climate emergency.
Fortunately, governments, businesses, and researchers are coming together to take on this astronomical challenge. In fact, in January 2021, the Japanese government declared that it will become ‘carbon neutral’ by 2050. One promising avenue being pursued to achieve this goal is the investment and research into renewable energy and carbon-free technologies.

“Advances in green technology are paramount to our future survival, and renewable energy from sources such as the sun, wind, and waves are excellent options,” explains Akari Hayashi, a professor at Kyushu University's Platform of Inter-/
“However, these energy sources cannot always match demand, so we need ways to store that energy. Rechargeable batteries work great for the short term, and for long term storage, we can convert that energy into hydrogen.”
Kyushu University has been a leader in hydrogen energy research, and Hayashi’s world revolves around improving the very heart of the technology itself: the electrocatalyst of the hydrogen fuel cell.
In its most basic form, hydrogen is an atom with one positively charged particle, a proton, and one negatively charged particle, an electron. When a hydrogen molecule enters a fuel cell, an electrocatalyst breaks apart the molecule into those core components.
The electrons pass through a circuit, thereby generating electricity, while the protons pass through a membrane to the other side of the fuel cell. When the proton and electron meet back up, they react with oxygen to produce water, the only ‘waste’ made by the process.
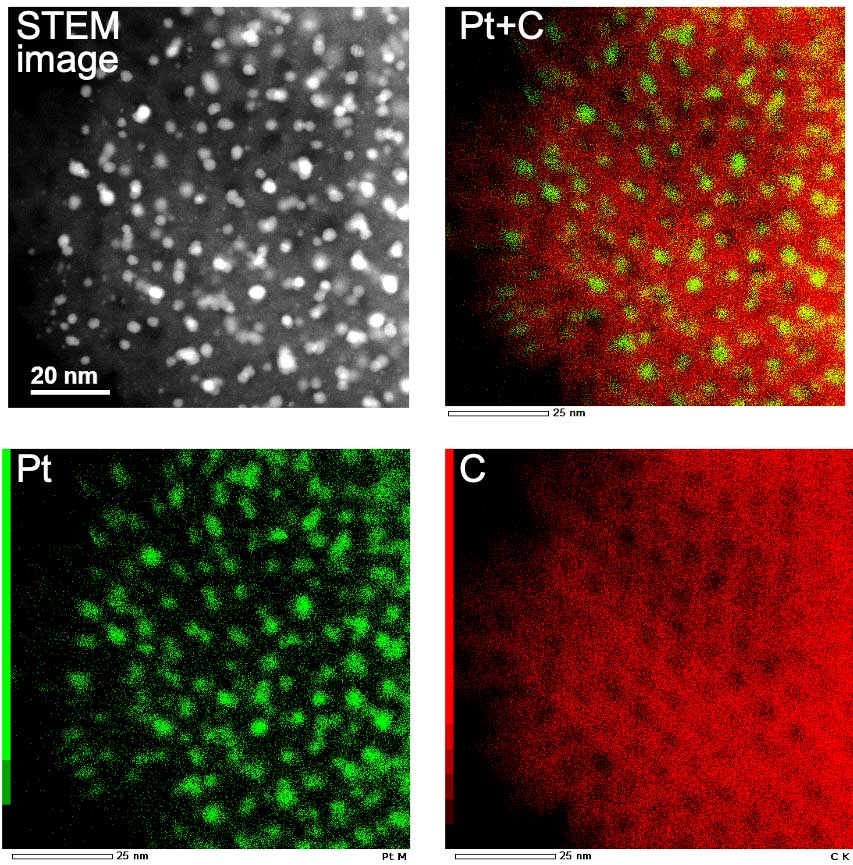
“The electrocatalyst speeds up all the chemical reactions that happen in the fuel cell,” says Hayashi. “They are made of a porous structure usually coated with a thin layer of platinum nanoparticles. Understanding the structure and organization of it is key to improving efficiency.”
Hayashi and her team have been working to understand and suppress electrocatalyst degradation and find ways to make electrocatalysts withstand higher operating temperatures.
“By studying how they degrade, we can begin to develop ways to prevent it so they can last longer,” continues Hayashi. “For example, platinum nanoparticles on the electrocatalyst can aggregate, leading to a reduction of surface area and reactivity. And if fuel cells can operate at higher temperatures without degrading, their utility in cars and larger vehicles would increase dramatically.”
Recently, her team has been developing ways to better produce hydrogen from renewable energy. Currently, hydrogen gas is commonly generated by splitting water molecules into hydrogen and oxygen—known as electrolysis—using electricity that often comes from fossil fuels. However, Hayashi’s team has shown promising results using wind power for the process.
Hayashi believes that the fundamental technology for fuel cells and electrolysis is mostly established. However, for a ‘hydrogen energy society’ to succeed, improvements in efficiency and utility are necessary, and better catalysts are just one piece of this puzzle.

“This technology has the potential to solve our world energy problem, but it must be supported with research and development of hydrogen infrastructure, combined with economic programs and comprehensive legislation” she explains. “Thus, I am also working to educate students so they can become outstanding researchers and engineers who can contribute to realizing this future.”
“Through the efforts happening around the world, we expect to see more robust expansion of fuel cell vehicles in the next ten years, and we hope that hydrogen will be used in larger vehicles such as ship and airplanes within 30 years,” concludes Hayashi.



































