研究成果 Research Results
- TOP
- News
- Research Results
- Dormant egg cells successfully obtained from mouse stem cells in the lab
Dormant egg cells successfully obtained from mouse stem cells in the lab
2019.06.04Research ResultsLife & Health
Researchers at Kyushu University’s Department of Stem Cell Biology and Medicine have uncovered the conditions necessary to successfully recreate dormant egg cells from mouse stem cells, providing useful insight for the further understanding and development of reproductive biology and medicine.
After birth, most of the egg cells in the ovaries of female mammals remain in a dormant state. These dormant eggs cells only mature later in life when needed, thereby ensuring a long period of fertility. While scientists studying mice have been able to create mature egg cells from stem cells, the egg cells created in the laboratory quickly pass through the dormant state and begin maturing, and the mechanism that keeps the eggs in the dormant state has been unclear.
“There are still many mysteries remaining about how egg cells are formed,” says So Shimamoto, the first author on the paper announcing the new results. “The environment in nature and in the lab is vastly different, so we have been searching for how to recreate the necessary conditions in the lab to obtain the same growth process found in nature.”
By comparing the various chemicals present at different stages in egg cells from mice to those in egg cells grown in the lab, the research group, led by Prof. Katsuhiko Hayashi, has now been able to identify some the key conditions necessary for recreating the dormant state.
First, the researchers found that they could partially reproduce the dormant state in the lab by inducing an increased amount of the protein FOXO3 in the egg cells, but this alone was not enough.
Further analysis indicated that egg cells formed in the mice showed signs of having been exposed to a reduced amount of oxygen. By limiting the oxygen reaching the egg cells in the lab while at the same time increasing the amount of FOXO3, the researchers obtained egg cells very similar to those in the dormant state from live mice.
“This is the closest so far that egg cells developed from stem cells in the lab have come to resembling those found in nature,” comments Prof. Hayashi. “We hope that the knowledge gained through this research will contribute to developments in reproductive medicine in the future.”
These findings provide a better picture of how various environmental conditions are involved in the mechanisms controlling some of the earliest steps of reproductive biology. The researchers are now searching for additional interactions and conditions that play roles in the development of egg cells to further improve this understanding.
For more information about this research, see “Hypoxia induces the dormant state in oocytes through expression of Foxo3,” So Shimamoto, Yohei Nishimura, Go Nagamatsu, Norio Hamada, Haruka Kita, Orie Hikabe, Nobuhiko Hamazaki, and Katsuhiko Hayashi, Proceedings of the National Academy of Sciences of the United States of America, (2019), https://doi.org/10.1073/pnas.1817223116
Release by William J. Potscavage Jr.
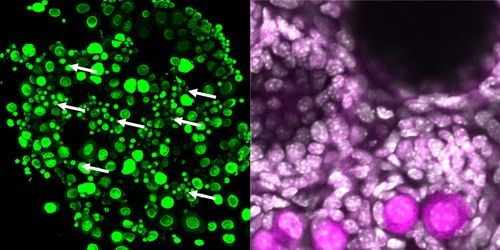
(Left image)
A reconstituted ovary created from embryonic stem cells with the egg cells treated to appear green. The arrows indicate induced dormant egg cells, which are much smaller than the larger, growing egg cells.
(Right image)
Dormant egg cells created from embryonic cells under oxygen-poor conditions. The protein FOXO3 is localized at the nuclei of the egg cells, which are relatively small.
Journal Reference
Hypoxia induces the dormant state in oocytes through expression of Foxo3, ,PNAS, https://doi.org/10.1073/pnas.1817223116Research-related inquiries
- TOP
- News
- Research Results
- Dormant egg cells successfully obtained from mouse stem cells in the lab































