研究成果 Research Results
- TOP
- News
- Research Results
- Discovery of novel proteins essential for DNA break repair
Discovery of novel proteins essential for DNA break repair
2018.11.01Research ResultsLife & Health
Immunodeficiency, centromeric instability, and facial anomalies (ICF) syndrome is an autosomal recessive disorder characterized by reduced immunoglobulin levels in the serum and recurrent infection. The patients’ chromosomes are fused via centromeric and/or pericentromeric regions, which are composed of repeat sequences.
In the present study, Dr. M. Unoki and Prof. H. Sasaki in the Medical Institute of Bioregulation, Kyushu University, and Prof. H. Funabiki in the Rockefeller University revealed that CDCA7 and HELLS proteins, which are mutated in ICF syndrome patients, are essential for non-homologous end joining (NHEJ), one of the major repair pathways of DNA breaks. Homologous recombination (HR), the alternative pathway, is unsuitable for repairing repeat sequences, because it includes a process of strand exchange between the DNA molecule with the same or similar sequences.
The research group speculates that, in ICF patients, a defect in NHEJ could lead to the distinctive chromosome configuration via the use of HR. Because defects in DNA repairs are the major triggers of carcinogenesis and mutations of proteins involved in NHEJ cause immunodeficiency, the research group believes that this study deepens the insights into these diseases.
This study was supported by JSPS KAKENHI (Grant numbers JP26253020 and JP18K06961), QR program of Kyushu University, and the Cooperative Research Project Program of the Medical Institute of Bioregulation, Kyushu University. The research achievement was published online in Journal of Clinical Investigation on Oct. 11, 2018.
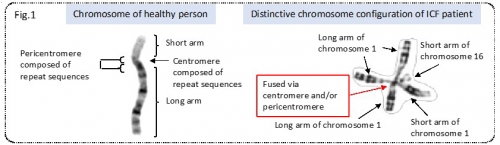
Figure 1. Representative chromosomes of healthy person and ICF syndrome patient.
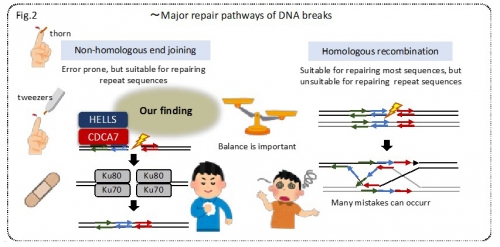
Figure 2. Schematic representation of major repair pathways of DNA breaks and our finding. The research group found that HELLS and CDCA7 proteins serve as a pair of tweezers to remove nucleosomes and help Ku80 to access and protect DNA break ends like a band-aid.
Journal Reference
CDCA7 and HELLS mutations undermine nonhomologous end joining in centromeric instability syndrome, ,The Journal of Clinical Investigation, 10.1172/JCI99751.Research-related inquiries
- TOP
- News
- Research Results
- Discovery of novel proteins essential for DNA break repair































