研究成果 Research Results
- TOP
- News
- Research Results
- DYNAMO1 drives division of mitochondria and peroxisomes by fuelling GTP
DYNAMO1 drives division of mitochondria and peroxisomes by fuelling GTP
2019.01.07Research ResultsLife & Health
Membrane fission including cell division is essential for the life of cells. Intracellular organelles such as mitochondria and peroxisomes proliferate by fission reactions mediated by the dynamin family of GTPase proteins such as Dnm1. These proteins polymerize and form a ring or spiral structure to constrict and pinch off the membrane by GTP hydrolysis energy generated by Dnm1. To address how a large amount of GTP is supplied to the division site, Professor Yukio Fujiki at the Medical Institute of Bioregulation, Kyushu University and JSPS research fellow Yuta Imoto, along with a team of researchers that include Assistant Professor Okumoto Kanji from the faculty of Science, Kyushu University and Visiting Professor Kuroiwa Tsuneyoshi from Japan Women's University used as a best model system a unicellular red alga, Cyanidioschyzon merolae, that contains a single each of mitochondrion, peroxisome, and plastid.
By proteomic analysis of highly purified mitochondrial division (MD) and peroxisome-dividing (POD) machinery, we for the first time identified the 17-kDa nucleoside diphosphate kinase-like protein, dynamin-based ring motive-force organizer 1 (DYNAMO1), and demonstrated that it locally generates GTP in MD and POD machineries. DYNAMO1 is widely conserved among eukaryotes up to humans and colocalizes with Dnm1 on the division machineries. DYNAMO1 converts ATP to GTP, and disruption of its activity impairs mitochondrial and peroxisomal fissions. DYNAMO1 forms a ring-shaped complex with Dnm1 and increases the constricting force. For the first time, their results identify DYNAMO1 as an essential component of MD and POD machineries, demonstrating that local generation of GTP in Dnm1-based machinery regulates motive force for membrane severance.
The paper, ‘Onsite GTP fuelling via DYNAMO1 drives division of mitochondria and peroxisomes’ has been published in Nature Communications. 9: article 4634 (2018)
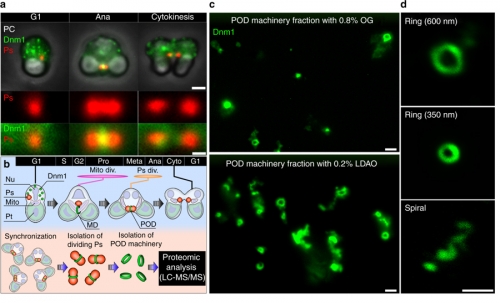
Figure 1. Proteomic analysis of POD machinery and identification of DYNAMO1.
Journal Reference
Onsite GTP fuelling via DYNAMO1 drives division of mitochondria and peroxisomes, ,Nature Communications, 10.1038/s41467-018-07009-zResearch-related inquiries
- TOP
- News
- Research Results
- DYNAMO1 drives division of mitochondria and peroxisomes by fuelling GTP































