研究成果 Research Results
- TOP
- News
- Research Results
- When oxygen attacks
When oxygen attacks
Genome-wide screening identifies novel cellular defense mechanisms against toxic and dangerous reactive oxygen species 2020.03.27Research ResultsLife & Health
While we all know that oxygen is essential for us to live, the cells in our bodies also have to defend themselves against highly reactive forms of oxygen found in some chemicals—including those produced naturally in our bodies—that can affect health and cause disease by damaging cells, DNA, and more if left unchecked.
A collaborative effort by researchers at Kyushu University and Stanford University now sheds new light on these defense mechanisms in a paper published in Cell Reports, highlighting potential genes that could be targeted in the future to treat or prevent certain diseases.
To find these new targets, the researchers used two techniques—called Cas9/CRISPR and shRNA screens—to modify hundreds of different genes in a type of human cell that can easily be grown and reproduced in the lab. While large samples of cells were all modified together, each cell only underwent a handful of the total possible genetic modifications because of the randomness of the process. Depending on the genes that were affected, different processes were disrupted in the cells.
The researchers then exposed the cells to cycles of hydrogen peroxide, a chemical containing highly reactive oxygen. This larger amount of reactive oxygen than the cells normally encounter causes a condition known as oxidative stress. Comparing the genetics of cells that did and did not survive upon hydrogen peroxide treatment, the researchers identified which genes were making the cells either more sensitive or more resistant to the reactive oxygen.
“While we found many relationships that were consistent with our understanding of different cell processes, we were surprised to find that disrupting genes known to play a role in some defense mechanisms actually improved the protection of the cells against oxidative stress,” says Yukio Fujiki, a member of Kyushu University’s Medical Institute of Bioregulation at the time of the work and a corresponding author on the paper reporting the results.
In particular, they found that deleting the genes involved in the import of proteins into a structure called the peroxisome, which plays a key role in regulating reactions like those caused by reactive oxygen, and in a process that is often activated as a first response to an abundance of reactive oxygen—called the pentose phosphate pathway—actually protected the cells.
In the former case, the researchers hypothesize that the improved survival is caused by an increase in the amount of catalase—an enzyme that can breakdown hydrogen peroxide—spread throughout the cell instead of localizing it to the peroxisome. In the latter case, the disruption appears to lead to the accumulation of a different molecule that then increases activity through the pathway.
“While these were just some of the surprises, the vast amount of new data collected through this study represents a rich resource for studying cellular response to oxidative stress going forward,” comments Fujiki.
For more information about this research, see “Systematic identification of regulators of oxidative stress reveals non-canonical roles for peroxisomal import and the pentose phosphate pathway,” Michael M. Dubreuil, David W. Morgens, Kanji Okumoto, Masanori Honsho, Kévin Contrepois, Brittany Lee-McMullen, Gavin McAllister Traber, Ria S. Sood, Scott J. Dixon, Michael P. Snyder, Yukio Fujiki, and Michael C. Bassik, Cell Reports (2020), https://doi.org/10.1016/j.celrep.2020.01.013
This study was supported in part by the Progress 100 project of Kyushu University, the Kyushu-U and Stanford-U Joint Research and Education Program, the Invitation Program for Top Global Researchers, grants from MEXT and JSPS (26116007, 15K21743, and 17H03675 to Y.F.), and the NIH Director’s New Innovator Award Program 1DP2HD084069 (to M.C.B.).
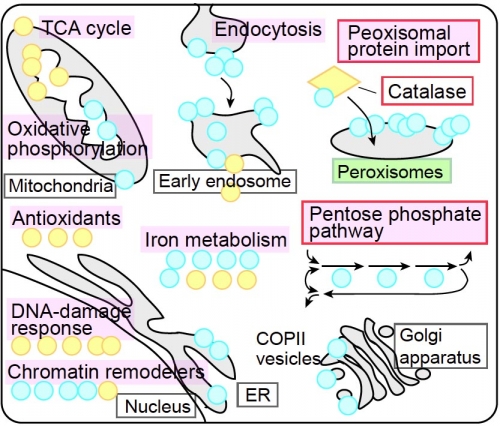
Figure 1. Schematic view of the pathways involved in oxidative stress response. Genome-wide screens identified multiple genes showing protective (blue) or sensitizing (yellow) effects against oxidative stress when respective genes were inactivated. Comprehensive analysis revealed that diverse pathways contribute to the cellular oxidative stress response (magenta). Peroxisomal protein import and pentose phosphate pathway showed strikingly potent and unexpected effects (red box).
Journal Reference
Systematic identification of regulators of oxidative stress reveals non-canonical roles for peroxisomal import and the pentose phosphate pathway, ,Cell Reports, https://doi.org/10.1016/j.celrep.2020.01.013Research-related inquiries
Yukio Fujiki, Principal Investigator, Joint Program with Faculty of Arts and Science
- TOP
- News
- Research Results
- When oxygen attacks































