研究成果 Research Results
- TOP
- News
- Research Results
- Understanding adhesion, one molecule at a time
Understanding adhesion, one molecule at a time
Researchers observe adhesion of a single polymer chain on a surface in their quest to develop an innovative adhesion technology 2021.03.11Research ResultsTechnologyEnvironment & Sustainability
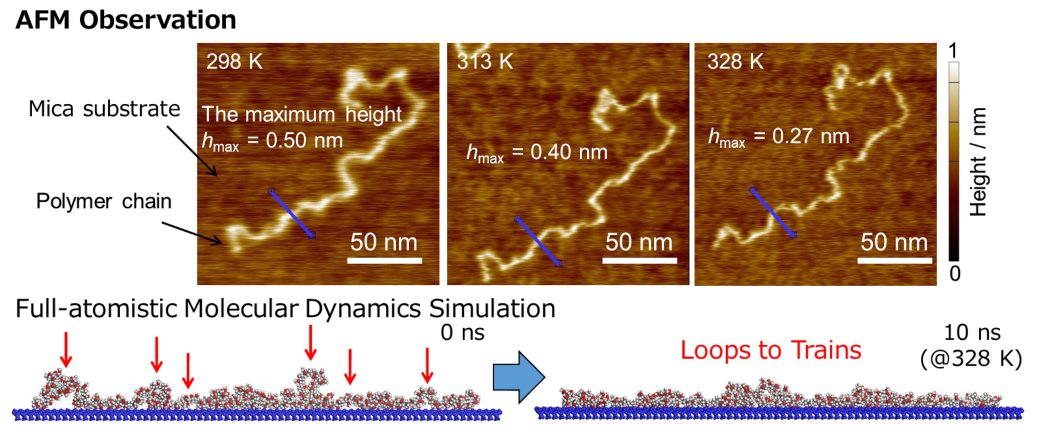
Researchers from Kyushu University have succeeded in directly observing the adhesion process for a single polymer chain on a surface and changes to its configuration upon heating. By understanding the adhesion process at a molecular level, the researchers hope to guide the design of stronger adhesives for plastics and improve lifetime prediction so the materials can play a bigger role in achieving light-weight vehicles for reduced energy consumption.
Conventionally, adhesion of a molecule to a surface, a process also known as adsorption, has been examined based mainly on indirect measurements using spectroscopy. However, a lack of a precise and comprehensive understanding for adhesion mechanisms at the molecular level is limiting the creation of adhesive technologies that can guarantee the strength and durability necessary for use in things such as all-plastic vehicles.
“Utilizing multi-materials or all-plastics for structural components is promising for reducing weight in next-generation vehicles to lower energy consumption and greenhouse gas emissions,” says Keiji Tanaka, leader of the study and professor in the Department of Applied Chemistry at Kyushu University’s Faculty of Engineering and the Center for Polymer Interface and Molecular Adhesion Science.
“Thus, we need to develop more robust adhesion technologies using polymeric materials, and a better understanding of how molecules adhere to a surface is an important step toward this goal.”
In the new work, Tanaka and his research team followed the change in configuration of polymer chains—long molecules like those found in plastics—as they adhered to a surface using atomic force microscopy, which allows for measurement of nanometer-size structures by basically dragging an extremely fine needle across a surface.
While the overall shape traced by a single polymer chain essentially remained unchanged with increasing temperature, the researchers observed decreases in the height and width of each chain, the images of which also became clearer.
“This indicates a transition from loops, or portions of the polymer chain that are not in contact with the surface, to trains, areas that are,” explains Yukari Oda, first author on the study. “Thus, we were able to observe the increase in contact points with the solid surface during the adsorption process.”
Furthermore, the researchers found that full-atomistic molecular dynamics simulations strongly supported the molecular picture obtained by atomic force microscopy.
“Such behaviors correspond to an elementary process for a solidification of adhesives on the surface,” states Tanaka.
###
For more information about this research, see “Direct observation of morphological transition for an adsorbed single polymer,” Yukari Oda, Daisuke Kawaguchi, Yuma Morimitsu, Satoru Yamamoto, and Keiji Tanaka, Scientific Reports 10, 20914 (2020). https://doi.org/10.1038/s41598-020-77761-0
This research was performed in collaboration with Prof. Satoru Yamamoto (Center for Polymer Interface and Molecular Adhesion Science), Prof. Daisuke Kawaguchi (Department of Applied Chemistry, Faculty of Engineering), and Dr. Yuma Morimitsu (Department of Applied Chemistry, Faculty of Engineering).
This research was a part of the “Innovative Adhesion Technology Based on 4-dimensional Multi-scale Analysis of Interfaces” project funded by the JST-Mirai Program. The projects aims to establish an innovative state-of-the-art adhesion technology based on in-situ 4-dimensional multi-scale analyses of adhered interfaces, in conjunction with molecular-level manipulation and fabrication of polymers. This project involves strong collaboration between the academe and industry and spans from establishing fundamental science at interfaces to manufacturing of components for mobility utilizing the envisioned adhesion technology.
This release is also available in Japanese.
Research-related inquiries
Keiji Tanaka, Professor
Department of Applied Chemistry, Faculty of Engineering Center for Polymer Interface and Molecular Adhesion Science
Contact information can also be found in the full release.
- TOP
- News
- Research Results
- Understanding adhesion, one molecule at a time































