研究成果 Research Results
- TOP
- News
- Research Results
- Development of New Catalytic Method for the Direct Synthesis of N-Unprotected α-Tertiary Amines
Development of New Catalytic Method for the Direct Synthesis of N-Unprotected α-Tertiary Amines
2017.05.01Research ResultsPhysics & Chemistry
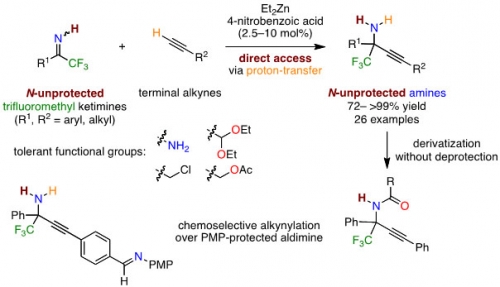
One of the most efficient methods to synthesize amines, which are ubiquitous in natural products and biologically active compounds, is nucleophilic addition to imines. In addition reactions, N-protected imines are often used because N-unprotected imines that could directly provide N-unprotected primary amines have the difficulties in the control of reactivity and stereoselectivity.
Professor Takashi Ohshima, Lecturer Hiroyuki Morimoto and Dr. Kazuhiro Morisaki from Kyushu University succeeded in developing new catalytic method: that is direct catalytic alkynylation reactions of N-unprotected trifluoromethyl ketimines, which directly affords N-unprotectedα-tetrasubstituted primary amines under proton-transfer conditions without additional deprotection steps. They also succeeded in direct transformation of the N-unprotected amine product into derivatives of biologically active compounds.
The above results have been published online on April 27, 2017 in Chemical Communications, an international journal from the Royal Society of Chemistry.
For more information about this research, see:
Direct access to N-unprotected tetrasubstituted propargylamines via direct catalytic alkynylation of N-unprotected trifluoromethyl ketimines.
K. Morisaki, H. Morimoto and T. Ohshima, Chem. Commun. 2017, Advance Article (DOI: 10.1039/c7cc02194a).
Journal Reference
Direct access to N-unprotected tetrasubstituted propargylamines via direct catalytic alkynylation of N-unprotected trifluoromethyl ketimines, ,Chemical Communications, 10.1039/c7cc02194aResearch-related inquiries
- TOP
- News
- Research Results
- Development of New Catalytic Method for the Direct Synthesis of N-Unprotected α-Tertiary Amines































