研究成果 Research Results
- TOP
- News
- Research Results
- Identification of a commitment factor to impact the generation of fatigue-resistant muscle
Identification of a commitment factor to impact the generation of fatigue-resistant muscle
2017.05.23Research ResultsLife & Health
The research group of Associate Professor Ryuichi Tatsumi clarified that the semaphorin 3A (Sema3A) ligand, which is secreted from resident myogenic stem satellite cells exclusively at the early-differentiation phase, impacts fatigue-resistant muscle fiber (slow-twitch fiber) generation through a signaling pathway: cell-membrane receptor (neuropilin2-plexinA3) → myogenin (and its cooperating regulators MEF2D/HDAC7) → slow myosin.
“We anticipate that fatigue-resistant fiber generation can be enhanced with agonists of the cell-membrane receptor (other than Sema3A), which have been already found in food ingredients as a promising natural agonist,” says Tatsumi.
This work on stem-cell biology has major implications for developing novel strategies in health sciences for promoting slow-twitch fiber expression in humans to combat sarcopenia and promote muscle endurance, and importantly for strategies to sustain food security through meat-animal production.
This research achievement was published on May 7, 2017 (US Eastern Standard Time), in the online edition of STEM CELLS (AlphaMed Press, Durham, NC, US).
For more information about this research, see Slow-Myofiber Commitment by Semaphorin 3A Secreted from Myogenic Stem Cells. DOI: 10.1002/anie.201507022R1
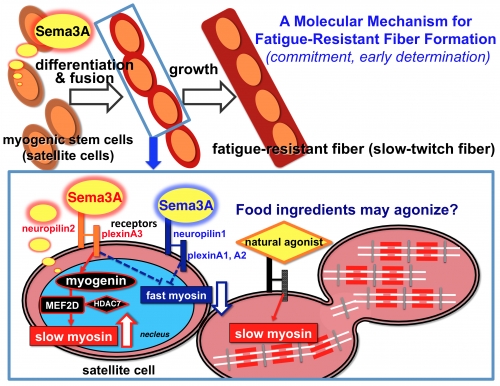
A model for a Sema3A-driven “commitment mechanism” to impact fatigue-resistant fiber (slow-twitch fiber) generation.
◆ Sema3A ligand is secreted from resident myogenic stem satellite cells exclusively at the early-differentiation phase (myofiber-formation phase).
◆ Sema3A binds to cell-membrane receptor neuropilin2-plexinA3 complex to impact slow-twitch fiber commitment through a signaling axis including myogenin, MEF2D, HDAC7, and slow myosin (red line).
◆ The model includes additional elements to suppress fast-myosin expression and hence enhance fatigue-resistant fiber formation (blue dashed-lines, projected from both neuropilin2-plexinA3 and neuropilin1-plexinA1, A2).
◆ It may be possible that food ingredients having agonistic activity of the receptor neuropilin2-plexinA3 can enhance the generation of fatigue-resistant fibers.

(Left)Assoc Prof. Mako Nakamura, Prof. Ryuichi Tatsumi, and Assist Prof. Wataru Mizunoya
Journal Reference
Slow-Myofiber Commitment by Semaphorin 3A Secreted from Myogenic Stem Cells, ,STEM CELLS, 10.1002/stem.2639Research-related inquiries
- TOP
- News
- Research Results
- Identification of a commitment factor to impact the generation of fatigue-resistant muscle































