研究成果 Research Results
- TOP
- News
- Research Results
- A new synthesis route for alternative catalysts of noble metals
A new synthesis route for alternative catalysts of noble metals
2017.08.03Research ResultsPhysics & Chemistry
Researchers have developed a new synthesis route for alternative catalysts of noble metals for versatile chemical reactions that could help address environmental concerns.
Noble metals such as platinum are useful as catalysts for versatile chemical reactions including fuel cell vehicles and reduction of CO2 emission. However, they are costly to be used for these purposes.
As inexpensive alternatives, organic-based catalysts and carbonaceous catalysts were explored, but both were ultimately found to be impractical. This was because organic-based catalysts tend to be active but unstable, while carbonaceous catalysts are stable but less active.
In this work, researchers believe to have found a solution by developing a new synthesis route for intermediate materials of organic-based catalysts and carbonaceous catalysts.
While conventional carbonaceous catalysts have amorphous carbonaceous structures that cause a decline in catalytic activities (Fig. 1a), the new synthesis route enables the formation of carbonaceous catalysts with controlled chemical structures like organic-based catalysts (Fig. 1b). This synthesis route is capable of developing alternative catalysts of noble metals for many eco-friendly technologies such as fuel cell vehicles, hydrogen generation from water and CO2 reduction.
“Network Joint Research Center for Materials and Devices” is a center to deepen and create new framework for the collaborative research among five different research institutes. This research was conducted through the “Five-Star Alliance research program” of the research center.
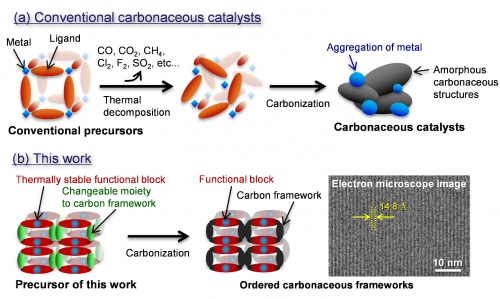
Fig. 1 Synthesis schemes of (a) conventional carbonaceous catalysts and (b) this work for ordered carbonaceous frameworks.
Journal Reference
Synthesis of ordered carbonaceous frameworks from organic crystals, ,Nature Communications , 10.1038/s41467-017-00152-zResearch-related inquiries
- TOP
- News
- Research Results
- A new synthesis route for alternative catalysts of noble metals































