研究成果 Research Results
- TOP
- News
- Research Results
- Electron doping improves photocatalytic activity for H2 and O2 evolution
Electron doping improves photocatalytic activity for H2 and O2 evolution
2018.07.02Research ResultsPhysics & ChemistryMaterialsTechnologyEnvironment & Sustainability
Dr. Yoshihiro Yamazaki at Kyushu University (Q-PIT), Dr. Kazuhiko Maeda at the Tokyo Institute of Technology and collaborators have found that the electron doping into SrTiO3 photocatalyst enlarges photocatalytic hydrogen and oxygen evolution reaction under UV light irradiation. The enhancement of hydrogen production was correlated well with an indication of prolonged lifetime of photoexcited electrons whereas that of oxygen evolution was due to the enlarged total flux of photoexcited holes towards the reaction site. These findings would lead to a new photocatalyst that could achieve higher efficiency in hydrogen production.
The article, entitled “Homogeneous Electron Doping into Non-Stoichiometric Strontium Titanate Improves Its Photocatalytic Activity for Hydrogen and Oxygen Evolution”, is published in ACS Catalysis as a just accepted article on 2018/6/19 (Tue). The definitive article will be available soon.
The work was carried out in collaboration with Kyushu University (Q-PIT), Tokyo Institute of Technology, Toyota Technical Institute and the National Institute for Materials Science (NIMS).
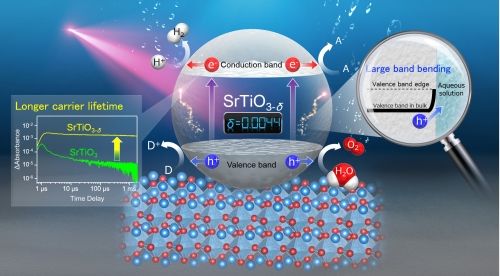
(Figure)
The lifetime of photoexcited electrons in electron-doped photocatalyst are significantly prolonged (left part in the figure). Electron doping, in addition, enlarges the surface band bending at solid-liquid interface (right part in the figure). Both significantly accelerate the photocatalytic H2 and O2 evolution reaction kinetics.
Journal Reference
Homogeneous Electron Doping into Nonstoichiometric Strontium Titanate Improves Its Photocatalytic Activity for Hydrogen and Oxygen Evolution, ,ACS Catalysis, 10.1021/acscatal.8b01379Research-related inquiries
Yoshihiro Yamazaki, Professor, Kyushu University Platform of Inter-/Transdisciplinary Energy Research (Q-PIT)
Kazuhiko Maeda, Associate Professor
Kazuhiko Maeda, Associate Professor
- TOP
- News
- Research Results
- Electron doping improves photocatalytic activity for H2 and O2 evolution































