研究成果 Research Results
- TOP
- News
- Research Results
- Making amino acids with electricity
Making amino acids with electricity
Highly efficient amino acid synthesis achieved from biomass-derivable acids and water using electric energy 2020.01.31Research ResultsPhysics & ChemistryTechnology
New research from Kyushu University could one day help provide humans living away from Earth some of the nutrients they need to survive in space or even give clues to how life started.
Researchers at the International Institute for Carbon-Neutral Energy Research reported a new process using electricity to drive the efficient synthesis of amino acids, opening the door for simpler and less-resource-intensive production of these key components for life.
In addition to being the basic building blocks of proteins, amino acids are also involved in various functional materials such as feed additives, flavor enhancers, and pharmaceuticals.
However, most current methods for artificially producing amino acids are based on fermentation using microbes, a process that is time and resource intensive, making it impractical for production of the vitals nutrients in space-limited and resource-restricted conditions.
Thus, researchers have been searching for efficient production methods driven by electricity, which can be generated from renewable sources, but efforts so far have used electrodes of toxic lead or mercury or expensive platinum and resulted in low efficiency and selectivity.
Takashi Fukushima and Miho Yamauchi now report in Chemical Communications that they succeeded in efficiently synthesizing several types of amino acids using abundant materials.
“The overall reaction is simple, but we needed the right combination of starting materials and catalyst to get it to actually work without relying on rare materials,” says Yamauchi.
The researchers settled on a combination of titanium dioxide as the electrocatalyst and an organic acid called alpha-keto acid as the key source material. Titanium dioxide is abundantly available on Earth, and alpha-keto acid can be easily extracted from woody biomass.
Placing the alpha-keto acid and a source of nitrogen, such as ammonia or hydroxylamine, in a water-based solution and running electricity through it using two electrodes, one of which was titanium dioxide, led to synthesis of the seven amino acids—alanine, glycine, aspartic acid, glutamic acid, leucine, phenylalanine, and tyrosine—with high efficiency and high selectivity even under mild conditions.
Hydrogen, which is also needed as part of the reaction, was generated during the process as a natural result of running electricity between electrodes in water.
In addition to demonstrating the reaction, the researchers also built a flow reactor that can electrochemically synthesize the amino acids continuously, indicating the possibilities for scaling up production in the future.
“We hope that our approach will provide useful clues for the future construction of artificial carbon and nitrogen cycles in space,” comments Yamauchi.
“Electrochemical processes are also believed to have played a role in the origin of life by producing fundamental chemicals for life through non-biological pathways, so our findings may also contribute to the elucidation of the mystery of the creation of life,” she adds.

Amino acid reactor
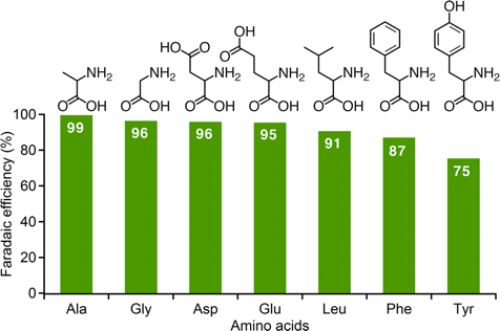
Fig. 1. Electrochemical production of various amino acids from the corresponding alpha-keto acids and hydroxylamine.
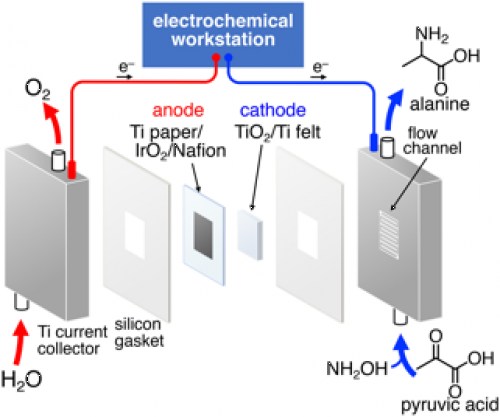
Fig. 2. A schematic image of a flow-type electrochemical reactor for the electrochemical amino acid synthesis.
Journal Reference
Electrosynthesis of amino acids from biomass-derivable acids on titanium dioxide, ,Chemical Communications (2019), https://doi.org/10.1039/c9cc07208jResearch-related inquiries
- TOP
- News
- Research Results
- Making amino acids with electricity































