研究成果 Research Results
- TOP
- News
- Research Results
- Zinc-Redox crosstalk: a new key to cellular protein quality control
 Zinc-Redox crosstalk: a new key to cellular protein quality control
Zinc-Redox crosstalk: a new key to cellular protein quality control
A newly discovered molecular link between zinc overload and protein misfolding offers fresh targets for therapeutic intervention
Professor Kenji Inaba
Medical Institute of Bioregulation
2026.04.28Research ResultsLife & Health
Fukuoka, Japan—Zinc is an essential trace element that controls myriad biological processes. The delicate balance of its concentration in the body is critical; both deficiency and excess are linked to severe pathological states such as impaired immunity, delayed wound healing, and sensory dysfunctions. However, it remains unclear how zinc dysregulation leads to these cellular defects at the molecular level.
In a study published in Nature Communications on April 22, 2026, researchers revealed that zinc levels in the endoplasmic reticulum (ER) control reduction-oxidation (redox) enzymes necessary for proper protein folding. Disruption in these enzymes directly impacts protein homeostasis, resulting in numerous pathologies.
The endoplasmic reticulum (ER) is the cell’s primary factory for synthesizing secretory proteins. For these newly synthesized proteins to function correctly, they must fold properly, a process that requires a tightly regulated intracellular redox environment.
“The motivation for this study stemmed from a key observation regarding how the cell partitions zinc,” explains Professor Kenji Inaba from Medical Institute of Bioregulation, Kyushu University. "We previously found that the zinc concentration in the ER is maintained at much lower levels than in the cytosol, the fluid inside the cell. This observation led us to question the physiological significance of low ER zinc levels and what might occur if that homeostasis was disrupted."
The team studied the role of ZIP7, a zinc transporter localized on the ER membrane. While ZIP7 deficiency was previously known to induce ER stress and developmental defects through an increase in zinc concentration in the ER, the precise molecular link between this transporter and protein quality control had remained elusive. To bridge this gap, the researchers developed techniques to visualize zinc ions within the ER of living cells and observed how ER zinc concentration changes following the inhibition of ZIP7.
Through a combination of in vitro biochemical assays and live-cell experiments, the team demonstrated that the ER zinc surge directly interferes with Ero1 activity. This factor is a crucial redox enzyme responsible for maintaining the oxidative environment necessary for protein “oxidative folding,” the essential process that stabilizes a protein’s three-dimensional structure.
This discovery reveals a sophisticated and previously unrecognized crosstalk between zinc levels and redox homeostasis. Building on these findings, the team inhibited Ero1 and showed that excess zinc stalls the oxidative folding of membrane proteins critical for cell signaling and growth, which can explain how cellular zinc imbalance may contribute to diseases such as cancer.
This research demonstrates for the first time the critical role zinc plays in governing the ER redox environment. The team hopes their methodology can extend to study other cellular components, such as the Golgi apparatus and lysosomes, to offer a new lens into how zinc affects the cell as a whole. Ultimately, these mechanistic insights provide a vital foundation for developing targeted therapeutic strategies to optimize zinc homeostasis and improve human health.
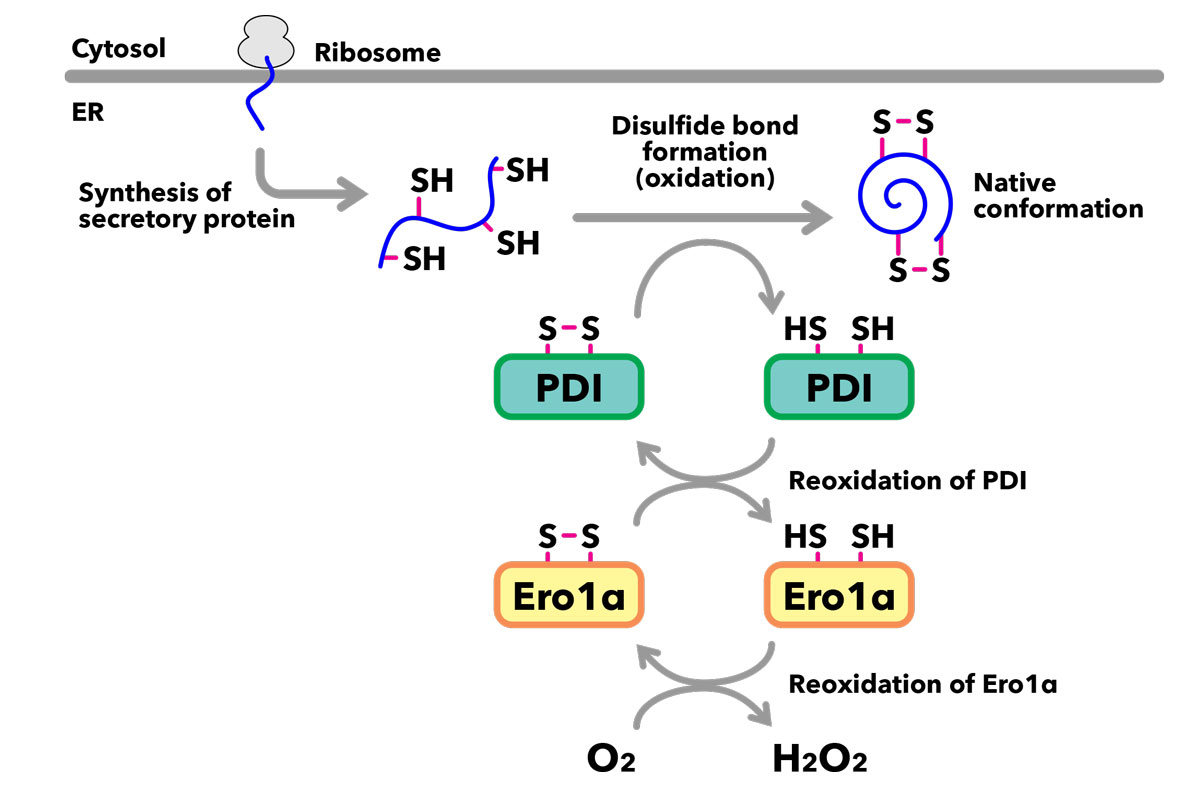
Newly synthesized secretory proteins are translocated into the ER, where disulfide bonds are formed between appropriate pairs of cysteine residues, enabling acquisition of their native conformations. Ero1α and PDI are central enzymes that catalyze this series of oxidation-reduction (redox) reactions.
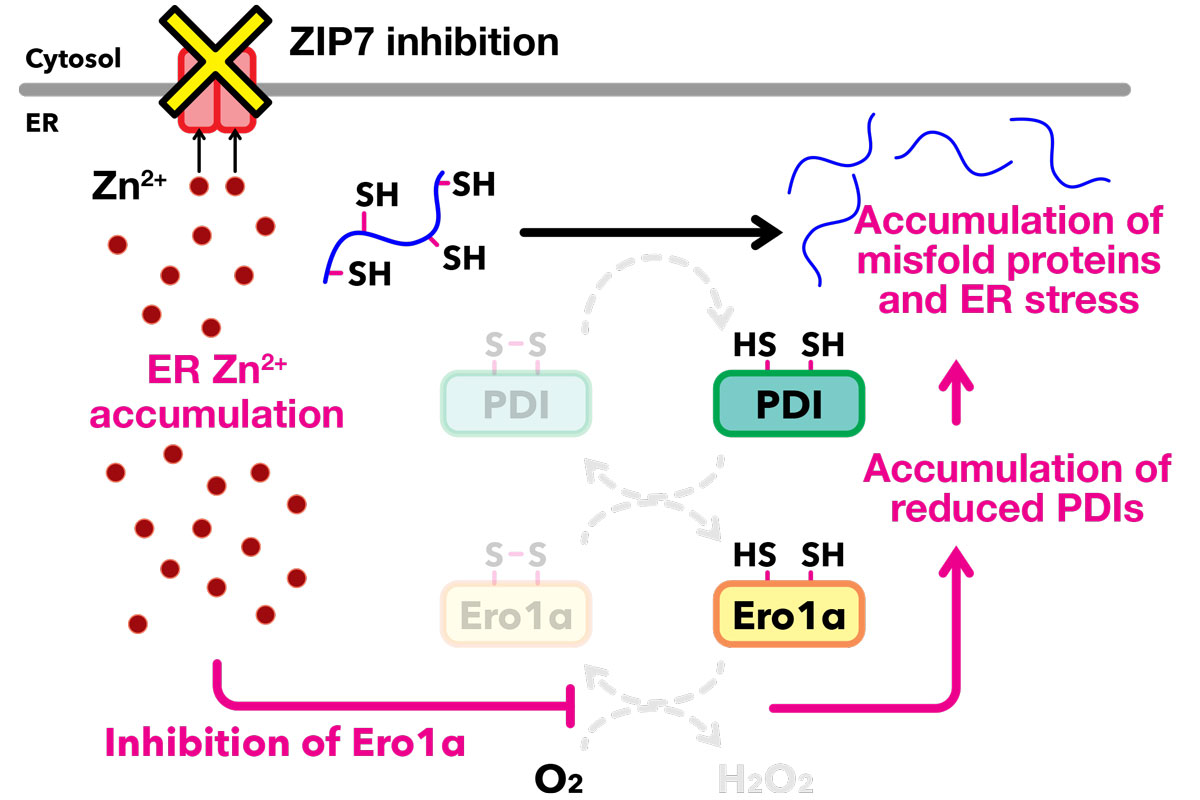
Inhibition of ZIP7, which localizes to the ER membrane, leads to the accumulation of Zn2+ within the ER. This excess Zn2+ directly inhibits the activity of Ero1α, thereby impairing the reoxidation of PDI. As a result, disulfide bond formation in newly synthesized secretory proteins is prevented, leading to the accumulation of misfolded proteins and the induction of ER stress.
###
For more information about this research, see "Zinc-redox crosstalk regulates proteostasis in the endoplasmic reticulum.” Yuta Amagai, Chihiro Arai, Wakana Yamamoto, Satoshi Watanabe, Toshiyuki Kowada, Roberto Sitia, Jun Hoseki, Shin Mizukami, Masaki Matsumoto, Kenji Inaba. Nature Communications, https://doi.org/10.1038/s41467-026-72250-w
Research-related inquiries
Kenji Inaba, Professor
Medical Institute of Bioregulation
Contact information can also be found in the full release.
- TOP
- News
- Research Results
- Zinc-Redox crosstalk: a new key to cellular protein quality control































